Crystal Structure of Q4D6Q6, a Conserved Kinetoplastid-Specific Protein From Trypanosoma Cruzi
É. Dias D'Andréa; Y. Roske*; G.A.P. de Oliveira; N. Cremer; A. Diehl; P. Schmieder; U. Heinemann; H. Oschkinat; J Ricardo Pires*
J. Struct .Biol. 211, 107563 (2020)
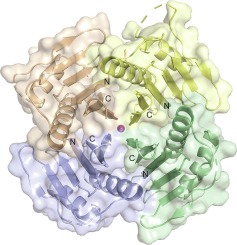
Complete genome sequencing of the kinetoplastid protozoans Trypanosoma cruzi, Trypanosoma brucei and Leishmania major (Tritryp), published in 2005, opened up new perspectives for drug development targeting Chagas disease, African sleeping sickness and Leishmaniasis, neglected diseases affecting millions of most economically disadvantaged people. Still, half of the Tritryp genes code for proteins of unknown function. Moreover, almost 50% of conserved eukaryotic protein domains are missing in the Tritryp genomes. This suggests that functional and structural characterization of proteins of unknown function could reveal novel protein folds used by the trypanosomes for common cellular processes. Furthermore, proteins without homologous counterparts in humans may provide potential targets for therapeutic intervention. Here we describe the crystal structure of the T. cruzi protein Q4D6Q6, a conserved and kinetoplastid-specific protein essential for cell viability. Q4D6Q6 is a representative of a family of 20 orthologs, all annotated as proteins of unknown function. Q4D6Q6 monomers adopt a ββαββαββ topology and form a propeller-like tetramer. Oligomerization was verified in solution using NMR, SAXS, analytical ultra-centrifugation and gel filtration chromatography. A rigorous search for similar structures using the DALI server revealed similarities with propeller-like structures of several different functions. Although a Q4D6Q6 function could not be inferred from such structural comparisons, the presence of an oxidized cysteine at position 69, part of a cluster with phosphorylated serines and hydrophobic residues, identifies a highly reactive site and suggests a role of this cysteine as a nucleophile in a post-translational modification reaction.
